This is the regulatory standard put in place by the United Staes Food and Drug Administration.
FDA APPROVED
The United States Food and Drug Administration (FDA) regulates three classes of food additives:
1: Direct food additives, such as food colouring, which are added to and remain in the food.
2: Secondary direct food additives, which are added to food during processing but are removed from the final product.
3: Indirect food additives, which are substances that come into contact with food but are not intended to be added to food. These additives can come from anything that food comes in contact with, including equipment, packaging, workwear, and personal protective equipment (PPE) such as work gloves.
The FDA food code recognises that various grades of work gloves are available for use by food facilities and classifies them as either single-use gloves or multiple-use gloves. This distinction is based on each glove type's durability, strength, and cleanability. Multiple-use gloves are durable, nonabsorbent, and resistant to corrosive food facility sanitisers. These gloves can withstand repeated washing and disinfecting treatments without being damaged or decomposing. Both multi- and single-use glove types must be safe for food contact, meaning there can be no migration of substance, colours, or tastes from or through the glove to the food being handled.
FDA Title 21 CFR Part 177 - Food and Drugs
In accordance with FDA regulations, gloves used in food service and food processing must comply with 21 CFR 177 parts 170-199, which specifies the requirement for items that are in repeated contact with food items and that are otherwise evaluated against indirect food additive regulations. FDA Title 21 CFR Part 177 states that the components of the glove must comply with applicable FDA regulations and must consist of “substances generally recognised as safe for use in food or food packaging.”
FDA 21 CFR Part 177.2600 - Rubber articles intended for repeated use
To ensure that gloves do not transfer harmful chemicals, colours, odours, or tastes to food, their material components must comply with FDA 21 CFR Part 177.2600. This Code of Federal Regulations (CFR) lists materials and chemicals that are permitted to come into contact with food. The CFR also details the accelerants, activators, antioxidants, plasticisers, fillers, and colourants that are acceptable for use in manufacturing these articles.
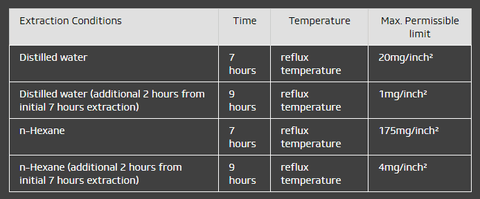
In addition, this code specifies the extractable limits for both fatty and aqueous (watery) foods and the testing, measuring (in milligrams per square inch), and reporting of overall migration (i.e., the total of the substances that can migrate). The testing is pass/fail.
Test Method table: (as specified in FDA 21 CFR Part 177.2600)
Extraction Conditions Time Temperature Max. Permissible limit
Distilled water 7 hours reflux temperature 20mg/inch²
Distilled water (additional 2 hours from initial 7 hours extraction) 9 hours reflux temperature 1mg/inch²
n-Hexane 7 hours reflux temperature 175mg/inch²
n-Hexane (additional 2 hours from initial 7 hours extraction) 9 hours reflux temperature 4mg/inch²